Asked Questions
How is Nectar linked to the Cytotron?
Heart Sanjivani Center’s Nectar The Cancer Institute as an irrevocable and perpetual license from Dr. Rajah Vijay Kumar, the inventor of the device, through his company Scalene Cybernetics Limited, India, for the marketing, manufacture and distribution of the Cytotron for degeneration and regeneration.
What does it mean by the Cytotron being granted a ‘Breakthrough device designation’ by the U.S. Food and Drug ADMINISTRATION (U.S.FDA)?
According to the FDA, the Breakthrough Devices Program is a program for certain medical devices and device-led combination products that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. The program is intended to help patients have more timely access to these medical devices by expediting their development, assessment, and review, while preserving the statutory standards for premarket approval, 510(k) clearance, and De Novo marketing authorization, consistent with the Agency’s mission to protect and promote public health. The FDA recognizes the Cytotron as a Breakthrough Device for the treatment of Breast, Pancreas and Liver Cancers, by conducting a clinical trial to establish that it is better than existing standards of care for these indications. This designation allows Shreis to Fast-track the path to market with FDA guidance, by conducting a clinical trial and establishing improvement in effectiveness of Cytotron therapy in these cancers. This breakthrough designation does not mean that the Cytotron is already an approved medical device for use in the US.
Who invented the Cytotron?
Rajah Vijay Kumar is the Group Chairman of Organization de Scalene, Director and Chief Scientific officer of the Centre for Advanced Research and Development (CARD), Director of Scalene Energy Research Institute (SERI) and Managing Director of Scalene Cybernetics limited and currently serves as Chairman of the board of Directors for over eight global organizations.
Is the Cytotron still experimental?
No. CYTOTRON therapy for the treatment of Musculoskeletal Disorders and terminal Cancer patients is not experimental. All required technical, safety and clinical requirements have been fulfilled to meet the device certification and approval criteria for the CE Mark (required Regulatory approval for clinical use of this technology in the European Union, like the US-FDA approval is required for clinical use in America. The Device is in clinical and commercial use in countries where the CE mark is adequate for treating patients. The Cytotron has been CE marked for clinical use since 2012. Domestic clinical trials and FDA approval is mandatory to get patients treated with the Cytotron here in this country. This being a platform technology, we are studying various disease indications with new protocols that are being developed fine tuned. Currently about 30 Cytotron installations are operating globally, for non-invasive tissue Degeneration (Cancer) and Regeneration (OA).
HOW much does the Cytotron treatment cost?
For accurate cost and pricing for treatment, please Contact Us for more information or the Cytotron treatment centers in your respective countries.
Is insurance reimbursement authorized for Cytotron treatment?
Insurance covers the entire cost of Cancer and OA treatment on the Cytotron in India. We are not aware of the situation in any other countries that have their own formularies. In the United States, once FDA approved clinical trials will be initiated soon. Nectar has every intention to have discussions with various medical insurance and treatment reimbursement companies to ensure inclusion in their formularies, for treatment with the Cytotron once approved.
See Our Latest Blog
- nectar
- 0 Comments
Cytotron Technology
A new ray of hope for Cancer and Osteo arthritis treatments Cytotron technology is to treat cancer and arthritis.Patients who come from across the world to avail this much better life, and most of them have survived for way longer than their doctors ever thought of. Many of them 61K their birthdays for years after […]
- nectar
- 0 Comments
What is Cytotron treatment
How does it work? At first look, you might confuse the Cytotron with a whole-body MRI scanner. However, it has a much bigger bore. Inside are guns that produce high instantaneous magnetic field and radio frequencies. These frequencies are specifically targeted at the problematic tissues. Our bodies have something called Trans-Membrane Potential pathways or TMP […]
- nectar
- 0 Comments
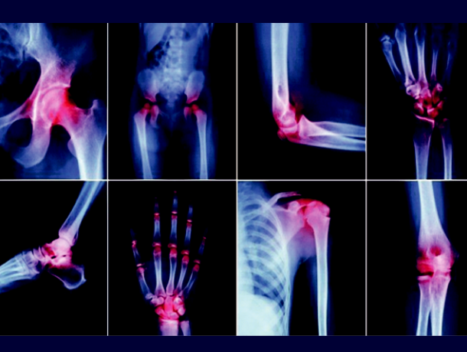
QMRT for Osteoarthritis
Quantum Magnetic Resonance Therapy (QMRT) for Osteoarthritis
- nectar
- 0 Comments
QMRT for Neoplastic Disorder
A globally approved medical device for tissue engineering.
- nectar
- 0 Comments
Tissue Degeneration – Cancer
We believe in providing a holistic treatment after a clinical evaluation by our specialized doctors and medical team.We suggest that you take a special MRI scan from our designated diagnostic centre, which will be used to customize the QMRT® programming and dosimetry to form your treatment schedule. Our standard osteoarthritis treatment is a 21-day programme, […]